UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, DC 20549
____________________
FORM 8-K
____________________
____________________
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of earliest event reported): September 3, 2025
(Exact name of registrant as specified in charter)
| (State or other jurisdiction | (Commission | (IRS Employer | ||||||||||||
| of incorporation) | File Number) | Identification No.) | ||||||||||||
(Address of principal executive offices) (Zip Code)
Registrant’s telephone number, including area code: (770 ) 651-9100
____________________
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Important Cautionary Statement
This report includes forward-looking statements. Statements regarding: (i) future sales or sales growth; (ii) our 2025 and longer term financial goals and expectations for future financial results, including levels of net sales, Adjusted EBITDA, Adjusted EBITDA margin, corporate expenses and cash; (iii) our expectations regarding the placental tissue market; (iv) our expectations regarding Medicare spending; and (v) continued growth in different care settings, are forward-looking statements. Additional forward-looking statements may be identified by words such as “believe,” “expect,” “may,” “plan,” “goal,” “outlook,” “potential,” “will,” “preliminary,” and similar expressions, and are based on management’s current beliefs and expectations.
Forward-looking statements are subject to risks and uncertainties, and the Company cautions investors against placing undue reliance on such statements. Actual results may differ materially from those set forth in the forward-looking statements. Factors that could cause actual results to differ from expectations include: (i) future sales are uncertain and are affected by competition, access to customers, patient access to healthcare providers, the reimbursement environment and many other factors; (ii) the Company may change its plans due to unforeseen circumstances; (iii) the results of scientific research are uncertain and may have little or no value; (iv) our ability to sell our products in other countries depends on a number of factors including adequate levels of reimbursement, market acceptance of novel therapies, and our ability to build and manage a direct sales force or third party distribution relationship; (v) the effectiveness of amniotic tissue as a therapy for particular indications or conditions is the subject of further scientific and clinical studies; and (vi) we may alter the timing and amount of planned expenditures for research and development based regulatory developments. The Company describes additional risks and uncertainties in the Risk Factors section of its most recent annual report and quarterly reports filed with the Securities and Exchange Commission. Any forward-looking statements speak only as of the date of this report and the Company assumes no obligation to update any forward-looking statement.
Item 7.01 Regulation FD Disclosure.
MiMedx Group, Inc. (the “Company”) is expected to participate in the Cantor Global Healthcare Conference on September 3, 2025 and has updated its investor presentation. A copy of the presentation materials is furnished as Exhibit 99.1 to this Current Report on Form 8-K (this “Current Report”) and is incorporated herein by reference.
The information in this Current Report, including Exhibits 99.1, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall such information be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, except as expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
| Exhibit No. | Description of Exhibit | |||||||
| 99.1 | ||||||||
| 104 | The cover page from this Current Report on Form 8-K, formatted in Inline XBRL. | |||||||
SIGNATURES
Pursuant to the requirements of the Exchange Act, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| MIMEDX GROUP, INC. | ||||||||
| September 3, 2025 | By: | /s/ Doug Rice | ||||||
| Doug Rice Chief Financial Officer | ||||||||

Cantor Global Healthcare Conference September 3, 2025 Investor Presentation

Disclaimer & Cautionary Statements This presentation and our earnings call includes forward-looking statements. Forward-looking statements are subject to risks and uncertainties, and the Company cautions investors against placing undue reliance on such statements. Actual results may differ materially from those set forth in the forward-looking statements. Such forward-looking statements include statements regarding: • Growing expansion outside of the U.S.; • Our growth expectations in 2025 and beyond, including our growth in surgery, increased funding in targeted research and expanded product portfolio; • Expected results of research and development, including that our efforts will innovate and diversify our product portfolio; • Placental-derived products and their potential clinical benefits; • Expectations regarding the reimbursement environment for the Company’s products, including Medicare Spending; • Expectations regarding HELIOGEN and AMNIOEFFECT driving Surgical growth; • CELERA’s impact on retaining business and its impact on our financial results; • Our expectations that we will continue to advocate for Medicare spending reform; • Exposure to tariffs and the anticipation that they will not impact the Company’s results; • 2025 full-year revenue growth and Adjusted EBITDA margin, our Long-term non-GAAP effective tax rates and top-line growth post reform in Medicare spending; • Our ability to manage Private Office dynamics, including adjusting our strategy to remain competitive; and • The Company’s long-term strategy and goals for value creation, the status of its pipeline products, expectations for future products, and expectations for future growth and profitability Additional forward-looking statements may be identified by words such as "believe," "expect," "may," "plan," "potential," "will," "preliminary," and similar expressions, and are based on management's current beliefs and expectations. Forward-looking statements are subject to risks and uncertainties, and the Company cautions investors against placing undue reliance on such statements. Actual results may differ materially from those set forth in the forward-looking statements. Factors that could cause actual results to differ from expectations include: • Future sales are uncertain and are affected by competition, access to customers, patient access to hospitals and healthcare providers, the reimbursement environment and many other factors; • The future market for the Company’s products can depend on regulatory approval of such products, which might not occur at all or when expected, and is based in part on assumptions regarding the number of patients who elect less acute and more acute treatment than the Company’s products, market acceptance of the Company’s products, and adequate reimbursement for such therapies; • The process of obtaining regulatory clearances or approvals to market a biological product or medical device from the FDA or similar regulatory authorities outside of the U.S. is costly and time consuming, and such clearances or approvals may not be granted on a timely basis, or at all, and the ability to obtain the rights to market additional, suitable products depends on negotiations with third parties which may not be forthcoming; and • The Company describes additional risks and uncertainties in the Risk Factors section of its most recent annual report and quarterly reports filed with the Securities and Exchange Commission. Any forward-looking statements speak only as of the date of this presentation and the Company assumes no obligation to update any forward-looking statement. Cantor Global Healthcare Conference – September 2025 2

A Pioneer and Leader Focused on Helping Humans Heal Cantor Global Healthcare Conference – September 2025 3 Over a decade of experience helping clinicians manage chronic and other hard-to-heal wounds Leading the industry with innovative products and robust supporting clinical data Poised to capitalize on favorable market trends to drive top line growth and profitability Our vision is to be the leading global provider of healing solutions through relentless innovation to restore quality of life.
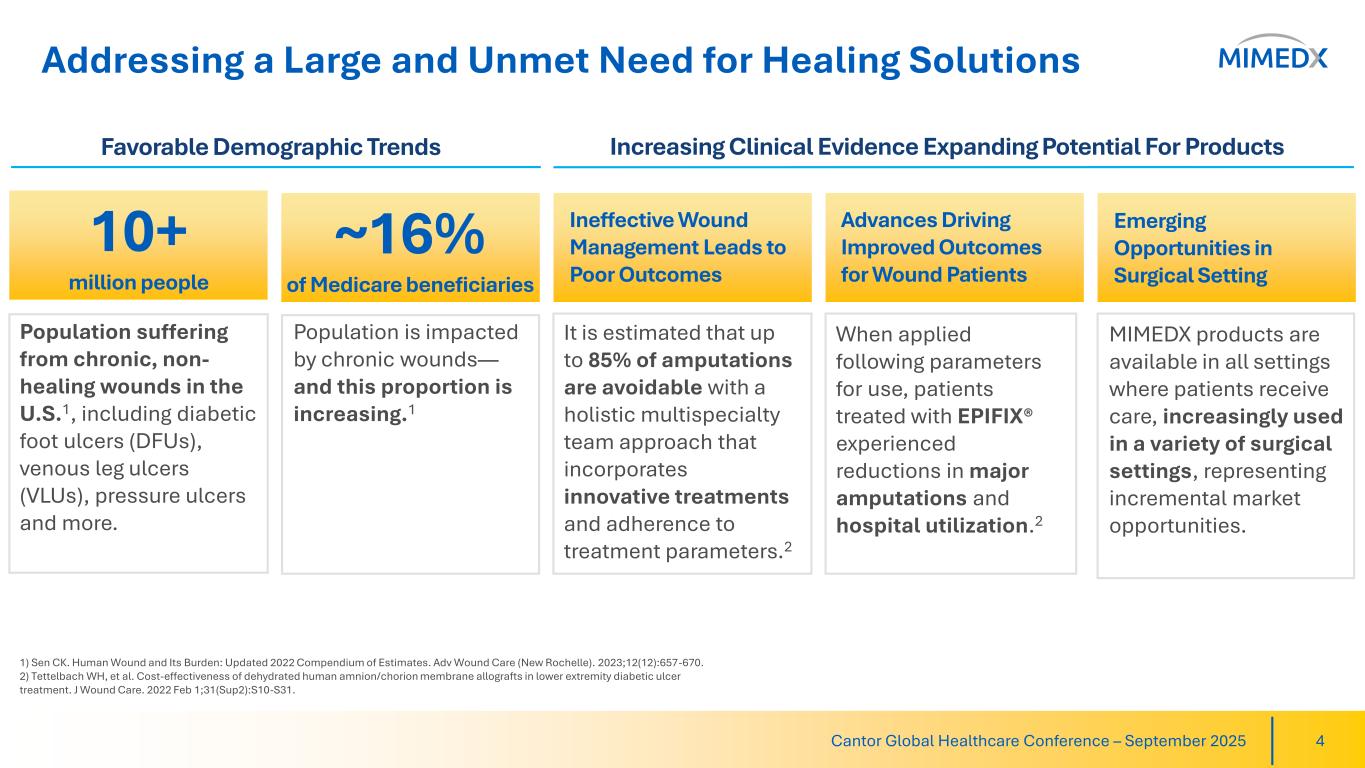
Addressing a Large and Unmet Need for Healing Solutions Cantor Global Healthcare Conference – September 2025 4 Favorable Demographic Trends Increasing Clinical Evidence Expanding Potential For Products 1) Sen CK. Human Wound and Its Burden: Updated 2022 Compendium of Estimates. Adv Wound Care (New Rochelle). 2023;12(12):657-670. 2) Tettelbach WH, et al. Cost-effectiveness of dehydrated human amnion/chorion membrane allografts in lower extremity diabetic ulcer treatment. J Wound Care. 2022 Feb 1;31(Sup2):S10-S31. Ineffective Wound Management Leads to Poor Outcomes When applied following parameters for use, patients treated with EPIFIX® experienced reductions in major amputations and hospital utilization.2 It is estimated that up to 85% of amputations are avoidable with a holistic multispecialty team approach that incorporates innovative treatments and adherence to treatment parameters.2 Population suffering from chronic, non- healing wounds in the U.S.1, including diabetic foot ulcers (DFUs), venous leg ulcers (VLUs), pressure ulcers and more. Population is impacted by chronic wounds— and this proportion is increasing.1 Advances Driving Improved Outcomes for Wound Patients Emerging Opportunities in Surgical Setting MIMEDX products are available in all settings where patients receive care, increasingly used in a variety of surgical settings, representing incremental market opportunities. 10+ million people ~16% of Medicare beneficiaries

Expansive Donor Network & IP Power Our Product Offering 5Cantor Global Healthcare Conference – September 2025 National Network of Birthing Center Partners Ample Placental Supply and Manufacturing Capabilities to Support Continued Growth and Industry Demand Expectant Mothers Introduced to Donation Program Consent for Donation Obtained Delivery of Healthy Baby via Caesarean Section Donated Placental Tissues Recovered Tissues Transported to MIMEDX Donor Tissue Tested & Prepared for Manufacturing Proprietary Processing Backed by Broad Portfolio of Intellectual Property Proprietary Processing & Terminal Sterilization of Tissues Shelf-Stable, Packaged Product Available to Ship Robust IP Estate with 200+ Patents Significant Opportunity for Continued Scale
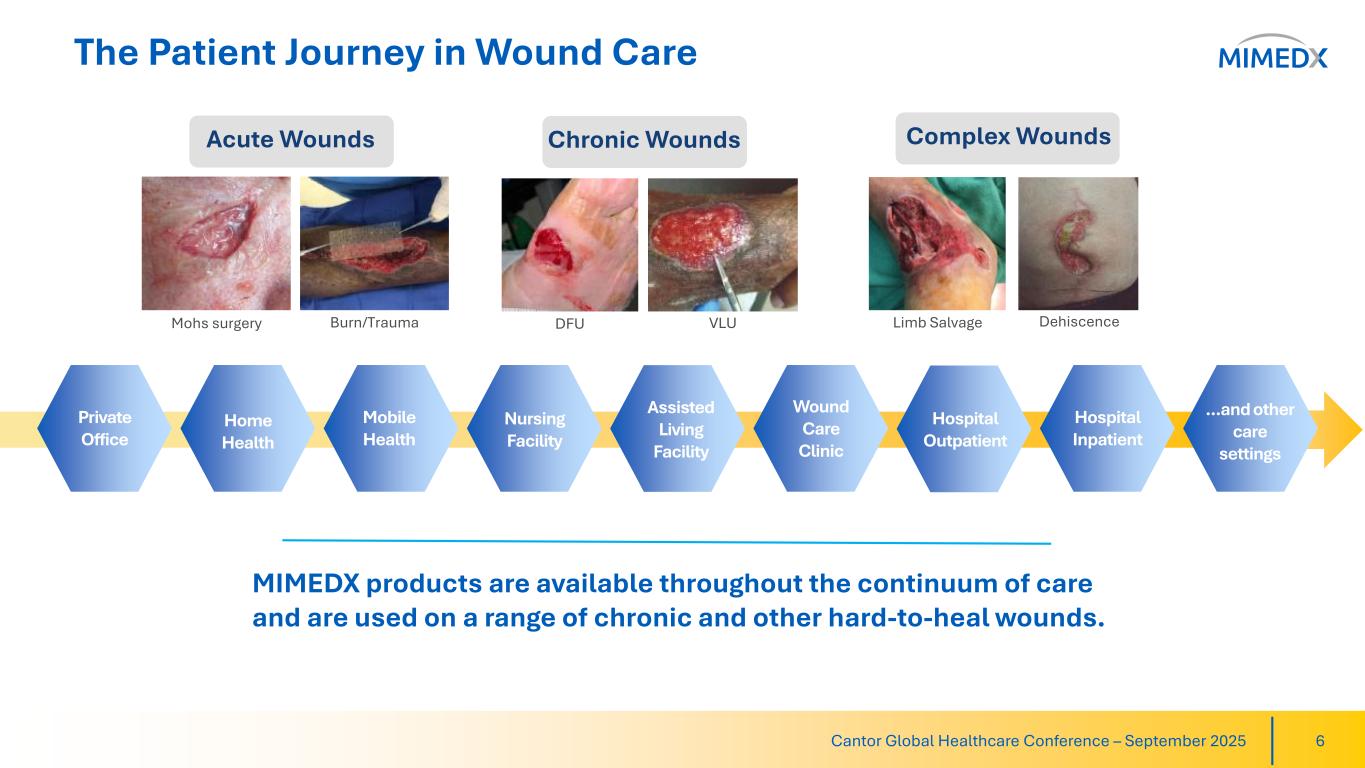
The Patient Journey in Wound Care 6Cantor Global Healthcare Conference – September 2025 Private Office Home Health Mobile Health Nursing Facility Assisted Living Facility Wound Care Clinic Hospital Outpatient Hospital Inpatient …and other care settings Mohs surgery Burn/Trauma DFU VLU Limb Salvage Dehiscence Acute Wounds Chronic Wounds Complex Wounds MIMEDX products are available throughout the continuum of care and are used on a range of chronic and other hard-to-heal wounds.

Surgical Studies Underway Highlight Product Versatility 7Cantor Global Healthcare Conference – September 2025 Published May 2025 Journal of Drugs in Dermatology Manuscript Pending RCT Enrollment Underway RCT Enrollment Underway Generating Clinical Data in Numerous Surgical Disciplines Incorporating Use of MIMEDX Products EPIFIX in Mohs (HECON) AMNIOFIX in GI Anastomosis AMNIOFIX in Liver Transplant AMNIOFIX in Breast Reduction AMNIOEFFECT in High-Risk Vascular E-Published April 2025 Vascular
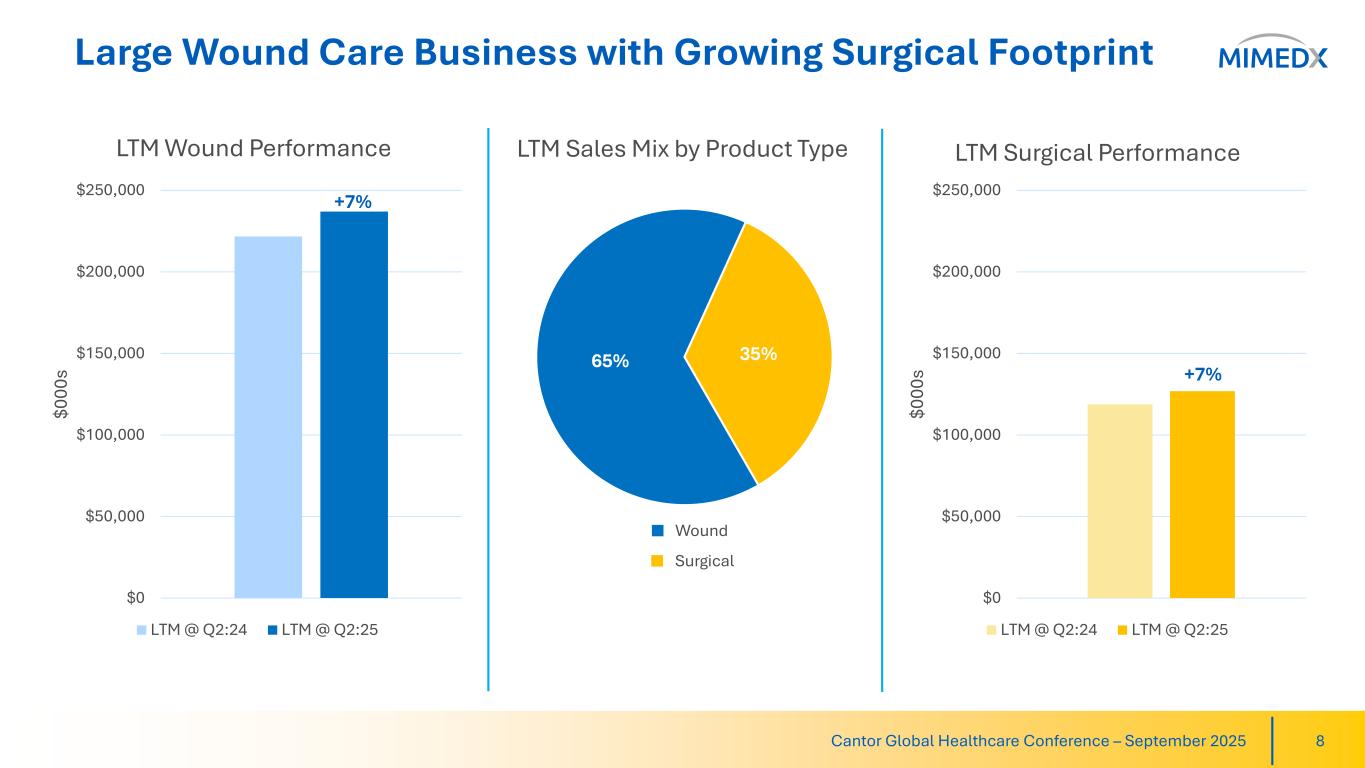
Large Wound Care Business with Growing Surgical Footprint 8Cantor Global Healthcare Conference – September 2025 $0 $50,000 $100,000 $150,000 $200,000 $250,000 $0 00 s LTM Surgical Performance LTM @ Q2:24 LTM @ Q2:25 $0 $50,000 $100,000 $150,000 $200,000 $250,000 $0 00 s LTM Wound Performance LTM @ Q2:24 LTM @ Q2:25 65% 35% LTM Sales Mix by Product Type Wound Surgical +7% +7%

Strategic Priorities Position Us to Win in 2025 & Beyond Investor Presentation – August 2025 9 Innovate & Diversify Product Portfolio to Maximize Growth Develop & Deploy Programs to Expand Surgical Footprint Enhance Customer Intimacy Enrollment continues for EPIEFFECT RCT with plans for interim report out Received TRG letter for our latest innovation, EPIXPRESS, clearing way for launch later this year Pilot programs underway for non-skin substitute Wound products, including with Vaporox Study on cost effectiveness of using MIMEDX products following Mohs surgery published in May in Journal of Drugs in Dermatology AMNIOFIX & AMNIOEFFECT continues to see strong uptake in surgical setting HELIOGEN sales accelerating with promising trajectory Evolving market dynamics place greater importance on comprehensive value offering, providing opportunity for MIMEDX Connect to drive stickiness Continue to develop programs and initiatives to further drive customer intimacy 1 2 3

Experienced, Skillful Leadership Team Executing Strategy Investor Presentation – August 2025 Prior Roles Include: Management Team with Track Record of Success in MedTech Joe Capper Chief Executive Officer Doug Rice Chief Financial Officer Butch Hulse Chief Administrative Officer & General Counsel Kim Moller Chief Commercial Officer Ricci Whitlow Chief Operating Officer John Harper, Ph.D. Chief Scientific Officer & SVP, R&D Matt Notarianni Head of IR 10
